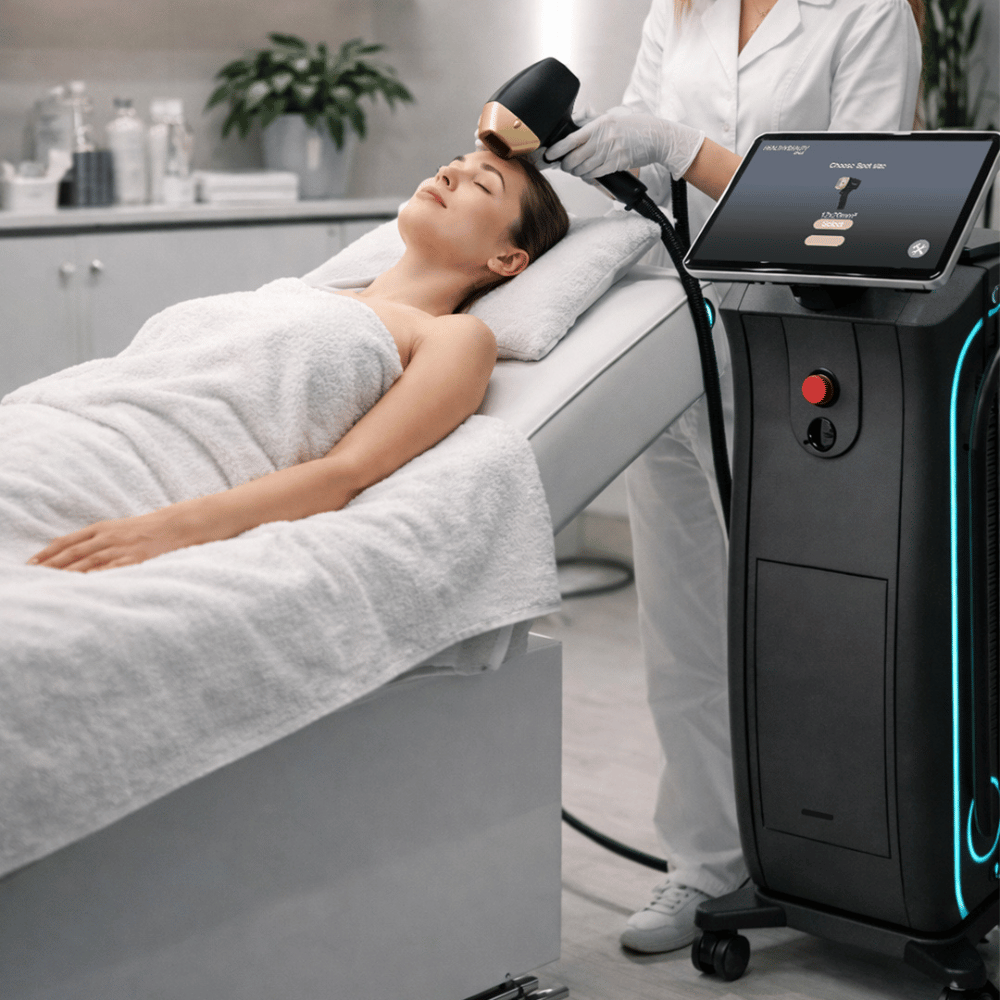
For clinics operating in North America, selecting an FDA-registered diode laser hair removal device enhances credibility and patient confidence.
Modern systems integrate:
-
diode laser hair removal 808nm
-
1064 diode laser hair removal
-
Triple wavelength diode laser 755nm 808nm 1064nm
Clinical literature supports diode laser epilation hair removal systems for long-term reduction when used under proper protocols (Sadick et al., 2000 – https://pubmed.ncbi.nlm.nih.gov/10886272/).
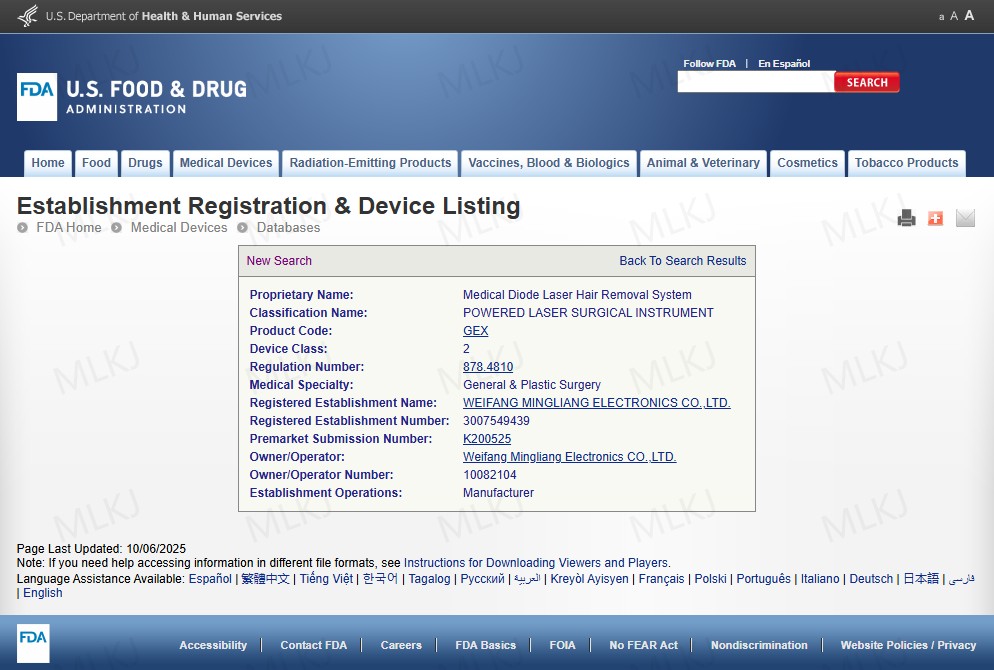
The diode laser epilator market continues to expand as clinics upgrade to professional 808nm diode laser hair removal device platforms.
Peer-reviewed research confirms safety across multiple phototypes, including diode laser hair removal for dark skin (Haedersdal et al., 2011 – https://pubmed.ncbi.nlm.nih.gov/21332911/).
Buyers comparing diode laser hair removal 808 and diode laser hair removal dark skin solutions increasingly demand FDA documentation before purchase.
Contact our compliance team to receive FDA-registered diode laser epilation equipment details.