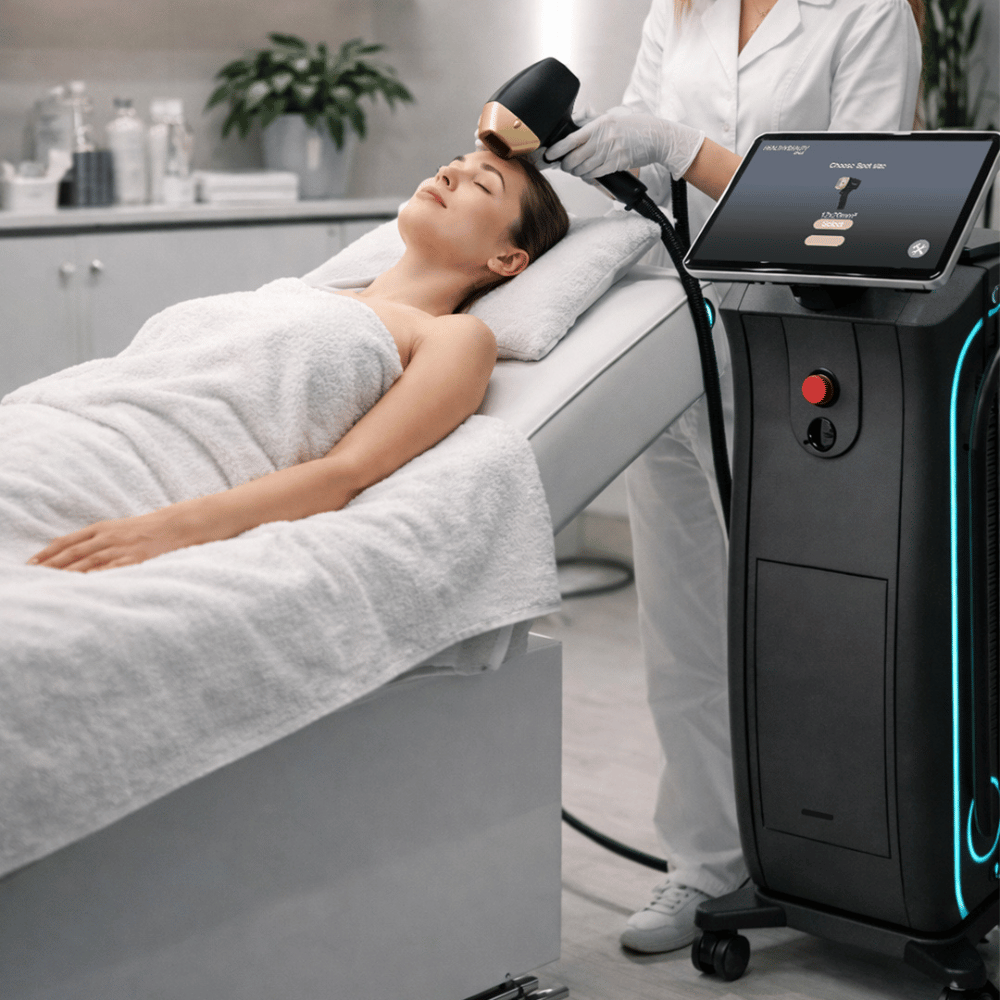
In the United States, investing in a diode laser hair removal machine requires more than comparing technical specifications. Clinics must evaluate compliance, safety, and long-term profitability. A laser hair removal machine that aligns with FDA standards provides operational security while supporting high patient volume.
Today’s leading professional hair laser removal machine integrates triple wavelength diode laser for hair removal technology. Often described as a triple wave diode laser, diode triple wave laser, or triple wavelength laser hair removal system, these platforms combine 808nm 755nm 1064nm diode laser outputs in one device. Configurations such as 755nm 808nm 1064nm diode laser or diode laser 755nm 808nm 1064nm offer versatility for different skin types and hair textures.
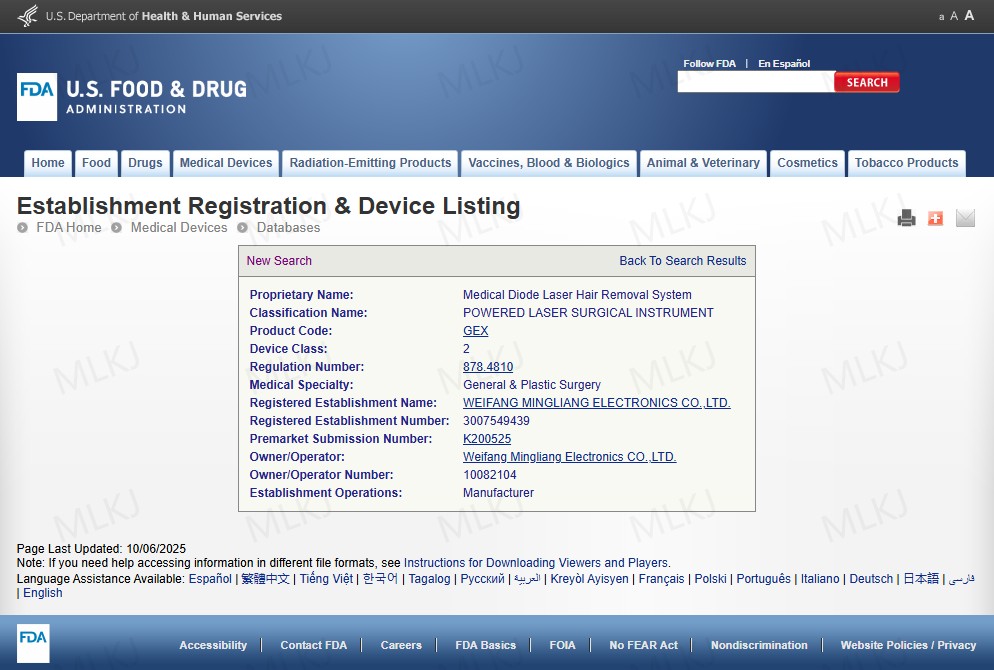
Clinical evidence continues to support 808nm hair removal as the backbone of diode-based treatment. A study published in Dermatologic Surgery confirmed sustained hair reduction with 808nm systems under appropriate parameters (Liew et al., 1999 – https://pubmed.ncbi.nlm.nih.gov/10534631/).
Even within 3 wavelength diode laser hair removal systems, the 808 diode laser machine remains central. High-quality 808nm laser diodes ensure energy stability, while advanced cooling supports triple ice laser diode hair removal comfort. A second clinical evaluation confirmed diode platforms provide predictable long-term outcomes when protocols are carefully followed (Alster & Tanzi, 2009 – https://pubmed.ncbi.nlm.nih.gov/19012262/).
As patient demographics diversify, the inclusion of 1064nm diode laser improves safety for darker skin types. Research has demonstrated diode systems maintain favorable safety profiles across multiple phototypes (Haedersdal & Wulf, 2006 – https://pubmed.ncbi.nlm.nih.gov/16489844/).
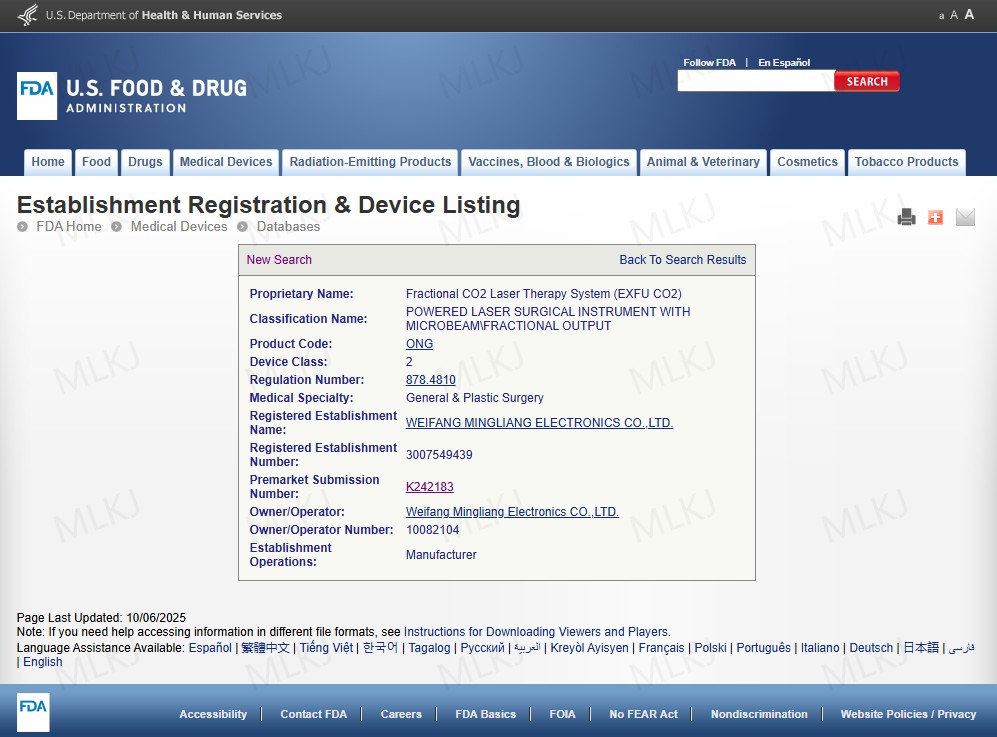
When comparing 808nm laser diode price and overall investment, U.S. clinics increasingly focus on durability, warranty, and regulatory documentation rather than initial cost alone. A compliant diode laser hair removal FDA approved device reduces liability and supports long-term growth.
Contact our FDA documentation team to receive full specifications and factory-direct pricing for your next-generation diode laser hair removal machine
For more information please contact us +86 18054417523